Our service hotline – we are here for you
Do you have questions about our products, orders, technical applications, or a project enquiry?
Please feel free to contact us by telephone. Our sales team will provide you with competent and reliable support.
📞 telephone number: +49 (0) 5121 7609 99
Availability:
Monday – Thursday: 8 a.m. to 5 p.m.
Friday: 8 a.m. to 4 p.m.
3% Shop Discount
When ordering products from the HEYL, HORIBA, JUMO und MILWAUKEE product portfolio via our online shop.
Shop Registration
Blog
HeylNeomeris as a system supplier also for under-counter systems in laboratories and medical practices
As a water purifier, we stand for disinfection and ultrapure water technology in particular. Our latest solution is particularly suitable for demineralized water requirements in laboratories and for operating medical practices to ensure that equipment and instruments are available at all times.
With a deionized water requirement of up to 60 litres/hour, our new system concept perfectly complies with the required limit values in accordance with guidelines 17 and 18 of the DGSV's Hygiene, Construction and Technology Committee in the smallest of spaces.
Hygiene and disinfection are important core objectives for every operator. The measurement of silicates and conductivity are components of our smart solution.
We support you in determining your demineralized water requirements and in smooth integration as a system supplier.
Trouble-free operation and the sustainable condition of equipment and instruments are essential for efficient process flow.
Water treatment plant as an under-counter system
What is a modular water treatment system for the outpatient surgical area?
The Neomeris modular water treatment system is a process water treatment system that provides ultrapure water for the reprocessing of surgical instruments and medical devices for the outpatient surgical area. It is used to treat the water in outpatient centers (MVZ - Medical Care Centers) on site so that it is suitable for medical purposes, especially sterilization.
The plant consists essentially in the standard configuration of 5 process stages and 2 additional optional process stages, which interlock and ensure the high purity of the water produced.
These include:
- the pre-filtration for the protection of the plant technology against possible particle entry from the supply network
- the activated carbon filter to protect the membrane technology used from damage by chlorine
- a variable capacity water softener, which can be used in the range from soft to hard water
- Reverse osmosis, which is suitable for small objects as well as for larger MVZs.
- a buffer tank for intermediate storage of the permeate produced by reverse osmosis
- a 2-stage mixed bed exchange system, which is monitored with regard to conductivity according to AKI and DGSV
- a recirculation section
- a water monitor with two sensors for monitoring the entire system with regard to leakages (in the event of any leakages, the inlet valve closes)
as well as
- connections for a bypass emergency supply.
Optionally, you can add the following modules depending on the area of application:
- Limit value measuring device type Testomat 808 SiO2 for monitoring the recommended silicate limit value of 0.4 mg/l in the ultrapure water produced
- a flow-through UV system (6th process stage)
- a pyrogen filter (7th process stage) and
- add the NeoTecMaster®, an open-manufacturer multicontroller system, to the system for location-independent monitoring of the measurement parameters. At the same time, the NeoTecMaster® enables visualization of the measured values at PC workstations when integrated into the customer's network structure.
Advantages
- + No limitation regarding input water hardness
- + Activated carbon filter for chlorine degradation
- + Highly scalable to 200-300 liters / hour (reverse osmosis)
- + Leakage sensor system for emergency shutdown
- + By-pass concept included in the overall plant concept
Optional advantages
- + Testomat 808 Silicate (avoidance of silicate slip, value retention of medical devices and instruments) - Plant and instrument protection
- + UV system (prevention of standstill germination)
- + Pyrogen filter (acts against fever-producing bacteria)
- + Lifting system (independence from the existing building services)
- + NeoTecMaster version (transmission of measured values to company network, company monitor) Integration into existing systems possible (multi-parameter concept open to manufacturers)


The water treatment plant for outpatient centers is designed with its plant size in such a way that it can fit into the smallest of spaces and, with its modular design, can be individually adapted to customer requirements.
The modular water treatment plant is an important component for the outpatient surgical area, as it ensures patient safety and the smooth functioning of medical equipment, as well as associated value preservation.
How does water treatment for sterile supplies and medical devices in outpatient centers work?
Water treatment for sterile supplies and medical devices in ambulatory surgical centers, such as eye clinics, is performed by a special water treatment system installed in the facility. The plant consists of the above components, which are capable of filtering, demineralizing and disinfecting the water available on site to ensure that it is suitable for sterilization of medical products.
Particular attention has been paid to the critical parameters of
- Chlorine: Chlorine-induced pitting corrosion is a frequently occurring damage event, which is detrimental to the service life of the instrumentation used and at the same time represents an initial source of further damage.
- Conductivity: As a cumulative parameter, conductivity is used to monitor the treated water with regard to the successful reduction or removal of all dissolved ionogens.
- Silicates: SiO2 forms a crystalline structure on the surfaces of the equipment and instrumentation, which can only be removed mechanically or chemically (hydrofluoric acid). Removal of the deposits results in the passivation layer not being able to form, which in turn is detrimental to the useful life of the instruments. If these deposits are not removed or even avoided, the resulting relief edges represent a kind of barrier on which critical substances can be deposited
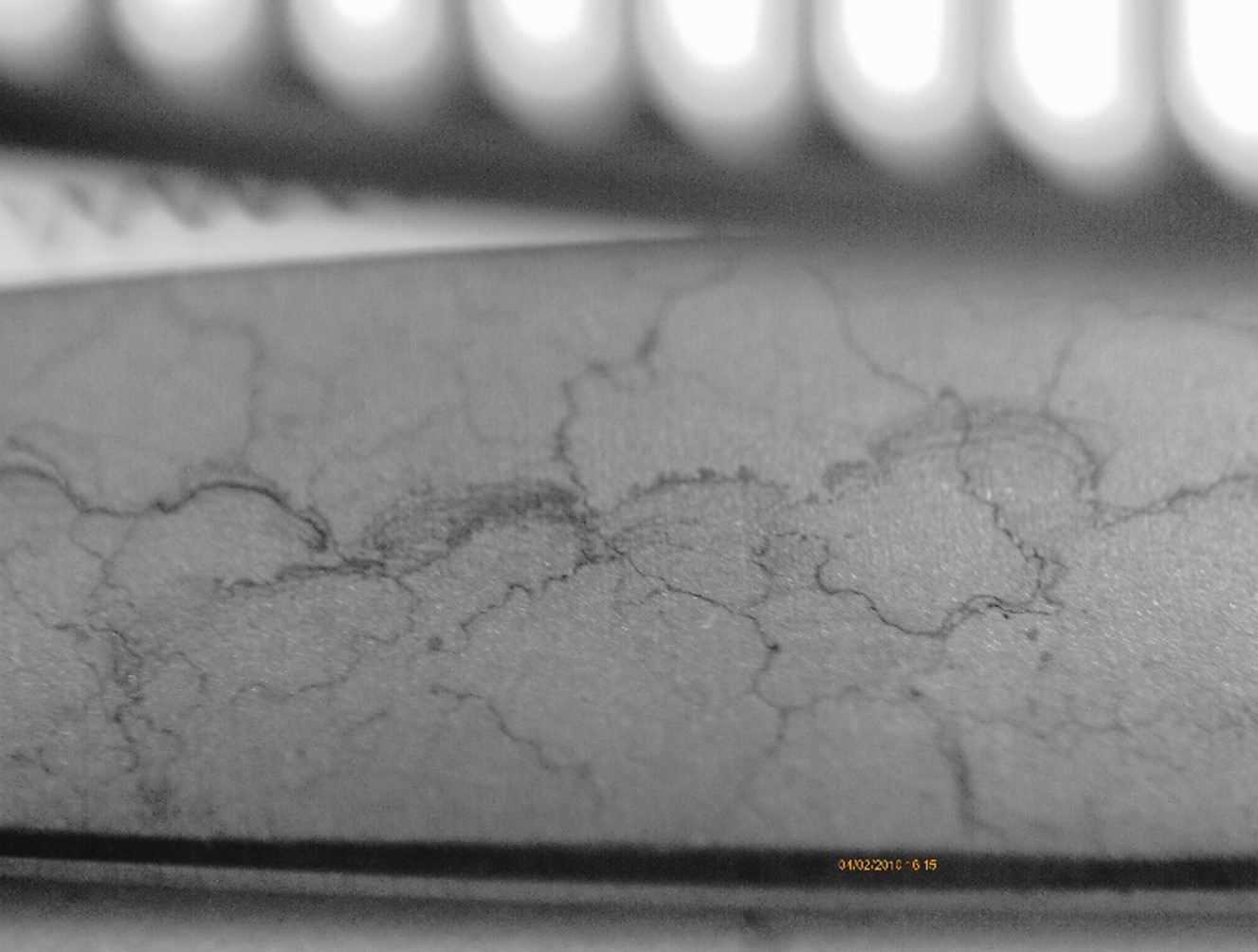

as well as
- Biological contaminants: Despite high-quality treatment of the water used, there is a residual risk of standstill contamination, especially during downtimes or improper operation of the water treatment system. To counteract this, a UV system with downstream pyrogen filter is installed in the recirculation section.
is installed.
The treated water is fed into the sterilization process to sterilize the medical devices and sterile supplies used in outpatient surgery. The quality of the treated water is regularly checked using suitable measuring equipment, e.g. the Testomat 808 Silicate. This ensures that the treated water complies with the applicable standards (DIN EN 285), recommendations of the AKI and the guidelines of the DGSV for compliance with the limit values for the reprocessing of safe medical devices and sterile goods and thus considers the issue of "patient safety" to the highest degree.
HeylNeomeris is a supporting member of the DGSV
(Deutsche Gesellschaft für Sterilgutversorgung e.V.)

Deutsche Gesellschaft für Sterilgutversorgung - DGSV® e.V.












